




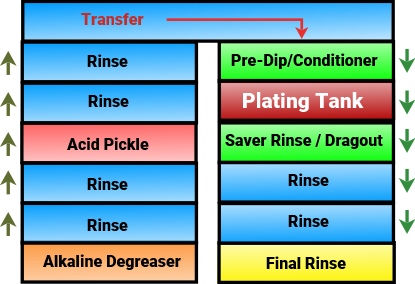
The DC power source referred to as the rectifier is shown at the top. The positive terminal leads to the anode (coloured red) and the negative terminal to the cathode (coloured blue)
The ions in solution are represented as spheres - Red spheres are the positively charged cations moving towards the cathode, blue coloured spheres are negatively charged anions moving towards the anode.
Cations convert to metal atoms when they reach the cathode and form the metallic deposit.
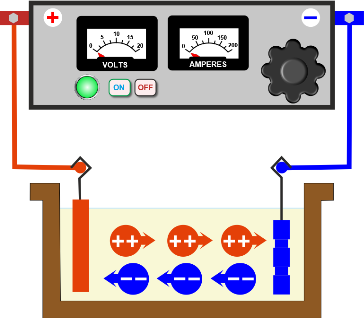


The primary components that have to be part of this tank are as follows:




During the plating process, a number of key parameters have to be managed. In a manual operation this requires careful oversight and continuous attention. On a busy line operators will be making regular adjustments to their solutions and ensuring that they are constantly aware of panel readings. Today, every function of the plating process can be automated. This includes:
